what is the maximum number of o_2 2 ​ molecules that can bind to a single molecule of hemoglobin?
This is the Wikipedia entry entitled "Hemocyanin". More...
The Wikipedia text that you see displayed here is a download from Wikipedia. This means that the information we display is a copy of the data from the Wikipedia database. The push button next to the article title ("Edit Wikipedia article") takes yous to the edit page for the article directly inside Wikipedia. You should be aware you are non editing our local copy of this data. Any changes that you lot make to the Wikipedia article volition not be displayed hither until we side by side download the commodity from Wikipedia. We currently download new content on a nightly footing.
Does Pfam agree with the content of the Wikipedia entry ?
Pfam has chosen to link families to Wikipedia articles. In some case we accept created or edited these articles simply in many other cases we accept not made whatever directly contribution to the content of the commodity. The Wikipedia community does monitor edits to try to ensure that (a) the quality of article annotation increases, and (b) vandalism is very speedily dealt with. However, we would like to emphasise that Pfam does not curate the Wikipedia entries and we cannot guarantee the accuracy of the information on the Wikipedia page.
Editing Wikipedia articles
Before y'all edit for the first time
Wikipedia is a free, online encyclopedia. Although anyone can edit or contribute to an article, Wikipedia has some potent editing guidelines and policies, which promote the Wikipedia standard of style and etiquette. Your edits and contributions are more likely to be accustomed (and remain) if they are in accordance with this policy.
You should take a few minutes to view the post-obit pages:
- Five pillars
- Policies and quidelines
- Wikipedia aid contents
- Wikipedia Tips
- Editing aid
How your contribution will exist recorded
Anyone can edit a Wikipedia entry. You can exercise this either as a new user or you tin can register with Wikipedia and log on. When you click on the "Edit Wikipedia commodity" button, your browser will direct you to the edit page for this entry in Wikipedia. If you are a registered user and currently logged in, your changes will exist recorded under your Wikipedia user proper noun. However, if you are not a registered user or are non logged on, your changes will be logged under your computer's IP address. This has 2 main implications. Firstly, every bit a registered Wikipedia user your edits are more likely seen as valuable contribution (although all edits are open up to community scrutiny regardless). Secondly, if you lot edit under an IP accost you may be sharing this IP address with other users. If your IP address has previously been blocked (due to being flagged every bit a source of 'vandalism') your edits volition also be blocked. You can find more information on this and creating a user business relationship at Wikipedia.
If yous have issues editing a particular page, contact united states of america at pfam-aid@sanger.ac.uk and we volition try to help.
Contact united states of america
The customs annotation is a new facility of the Pfam web site. If yous have problems editing or experience problems with these pages please contact us.
Hemocyanin Edit Wikipedia article
| Hemocyanin, copper containing domain | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
 Unmarried oxygenated functional unit of measurement from the hemocyanin of an octopus | |||||||||||
| Identifiers | |||||||||||
| Symbol | Hemocyanin_M | ||||||||||
| Pfam | PF00372 | ||||||||||
| InterPro | IPR000896 | ||||||||||
| PROSITE | PDOC00184 | ||||||||||
| SCOP2 | 1lla / SCOPe / SUPFAM | ||||||||||
| |||||||||||
| Hemocyanin, all-blastoff domain | |||||||||
|---|---|---|---|---|---|---|---|---|---|
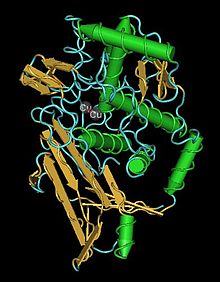
 Crystal structure of hexameric haemocyanin from Panulirus interruptus refined at 3.2 angstroms resolution | |||||||||
| Identifiers | |||||||||
| Symbol | Hemocyanin_N | ||||||||
| Pfam | PF03722 | ||||||||
| InterPro | IPR005204 | ||||||||
| PROSITE | PDOC00184 | ||||||||
| SCOP2 | 1lla / SCOPe / SUPFAM | ||||||||
| |||||||||
| Hemocyanin, ig-like domain | |||||||||
|---|---|---|---|---|---|---|---|---|---|
 crystallographic analysis of oxygenated and deoxygenated states of arthropod hemocyanin shows unusual differences | |||||||||
| Identifiers | |||||||||
| Symbol | Hemocyanin_C | ||||||||
| Pfam | PF03723 | ||||||||
| InterPro | IPR005203 | ||||||||
| PROSITE | PDOC00184 | ||||||||
| SCOP2 | 1lla / Telescopic / SUPFAM | ||||||||
| |||||||||
Hemocyanins (also spelled haemocyanins and abbreviated Hc) are proteins that transport oxygen throughout the bodies of some invertebrate animals. These metalloproteins incorporate two copper atoms that reversibly bind a single oxygen molecule (O2). They are 2d only to hemoglobin in frequency of use as an oxygen ship molecule. Unlike the hemoglobin in ruby-red blood cells found in vertebrates, hemocyanins are non confined in blood cells simply are instead suspended straight in the hemolymph. Oxygenation causes a color alter between the colorless Cu(I) deoxygenated course and the blue Cu(II) oxygenated course.[1]
Species distribution
Hemocyanins are found only in the Mollusca and Arthropoda: the earliest discoveries of hemocyanins were in the snail Helix pomatia (a mollusc) and in the horseshoe crab (an arthropod). They were subsequently found to exist mutual among cephalopods and crustaceans and are utilized by some country arthropods such as the tarantula Eurypelma californicum,[2] the emperor scorpion,[3] and the centipede Scutigera coleoptrata. Also, larval storage proteins in many insects appear to exist derived from hemocyanins.[4]
The hemocyanin superfamily
The arthropod hemocyanin superfamily is composed of phenoloxidases, hexamerins, pseudohemocyanins or cryptocyanins, and (dipteran) hexamerin receptors.[5]
Phenoloxidase are copper containing tyrosinases. These proteins are involved in the process of sclerotization of arthropod cuticle, in wound healing, and humoral immune defense. Phenoloxidase is synthesized past zymogens and are activated by cleaving an N-final peptide.[6]
Hexamerins are storage proteins commonly found in insects. These proteins are synthesized by the larval fat torso and are associated with molting cycles or nutritional conditions.[7]
Pseudohemocyanin and cryptocyanins genetic sequences are closely related to hemocyanins in crustaceans. These proteins have a similar construction and function, only lack the copper binding sites.[eight]
The evolutionary changes within the phylogeny of the hemocyanin superfamily are closely related to the emergence of these unlike proteins in diverse species. The understanding of proteins within this superfamily would non be well understood without the extensive studies of hemocyanin in arthropods.[9]
Structure and mechanism

The underside of the carapace of a red rock crab (Cancer productus). The regal coloring is acquired by hemocyanin.
Although the respiratory function of hemocyanin is like to that of hemoglobin, there are a significant number of differences in its molecular structure and machinery. Whereas hemoglobin carries its iron atoms in porphyrin rings (heme groups), the copper atoms of hemocyanin are bound as prosthetic groups coordinated by histidine residues. The active site of hemocyanin is composed of a pair of copper(I) cations which are directly coordinated to the protein through the driving forcefulness of imidazolic rings of six histidine residues.[10] It has been noted that species using hemocyanin for oxygen transportation include crustaceans living in common cold environments with depression oxygen pressure. Under these circumstances hemoglobin oxygen transportation is less efficient than hemocyanin oxygen transportation.[11] Nevertheless, there are as well terrestrial arthropods using hemocyanin, notably spiders and scorpions, that live in warm climates. The molecule is conformationally stable and fully functioning at temperatures upwards to ninety degrees C.[12]
Most hemocyanins demark with oxygen non-cooperatively and are roughly one-fourth as efficient equally hemoglobin at transporting oxygen per amount of blood. Hemoglobin binds oxygen cooperatively due to steric conformation changes in the protein complex, which increases hemoglobin's affinity for oxygen when partially oxygenated. In some hemocyanins of horseshoe crabs and some other species of arthropods, cooperative binding is observed, with Hill coefficients of 1.6â€"3.0. Hill coefficients vary depending on species and laboratory measurement settings. Hemoglobin, for comparison, has a Loma coefficient of ordinarily 2.8â€"3.0. In these cases of cooperative binding hemocyanin was bundled in protein sub-complexes of 6 subunits (hexamer) each with one oxygen bounden site; bounden of oxygen on 1 unit in the circuitous would increment the analogousness of the neighboring units. Each hexamer complex was arranged together to form a larger complex of dozens of hexamers. In one report, cooperative bounden was found to be dependent on hexamers being bundled together in the larger complex, suggesting cooperative bounden between hexamers. Hemocyanin oxygen-binding profile is also affected by dissolved salt ion levels and pH.[13]
Hemocyanin is made of many individual subunit proteins, each of which contains two copper atoms and can bind one oxygen molecule (O2). Each subunit weighs about 75 kilodaltons (kDa). Subunits may be arranged in dimers or hexamers depending on species; the dimer or hexamer complex is likewise arranged in chains or clusters with weights exceeding 1500 kDa. The subunits are usually homogeneous, or heterogeneous with two variant subunit types. Because of the large size of hemocyanin, it is usually found free-floating in the blood, unlike hemoglobin.[fourteen]
Hexamers are characteristic of arthropod hemocyanins.[fifteen] A hemocyanin of the tarantula Eurypelma californicum[ii] is made up of four hexamers or 24 peptide chains. A hemocyanin from the house centipede Scutigera coleoptrata[16] is made up of 6 hexamers or 36 bondage. Horseshoe venereal have an 8-hexamer (i. e. 48-chain) hemocyanin. Simple hexamers are found in the spiny lobster Panulirus interruptus and the isopod Bathynomus giganteus.[15] Peptide chains in crustaceans are well-nigh 660 amino acid residues long, and in chelicerates they are near 625. In the large complexes at that place is a diverseness of variant chains, all about the aforementioned length; pure components exercise not unremarkably self-assemble.[commendation needed]
Catalytic activity
Hemocyanin is homologous to the phenol oxidases (e.grand. tyrosinase) since both proteins share type 3 Cu active site coordination.[17] In both cases inactive proenzymes such as hemocyanin, tyrosinase, and catcholoxidase must be activated showtime. This is done by removing the amino acid that blocks the entrance channel to the agile site when the proenzyme is not active. There is currently no other known modifications necessary to activate the proenzyme and enable catalytic action. Conformational differences decide the type of catalytic activity that the hemocyanin is able to perform.[18] Hemocyanin also exhibits phenol oxidase activity, but with slowed kinetics from greater steric bulk at the active site. Partial denaturation really improves hemocyanin's phenol oxidase activity by providing greater admission to the active site.[i] [17]
Spectral properties

A hemocyanin agile site in the absenteeism of Oii (each Cu heart is a cation, charges not shown).

O2-bound course of a hemocyanin active site (the Cu2 heart is a dication, charge not shown).
Spectroscopy of oxyhemocyanin shows several salient features:[xix]
- Resonance Raman spectroscopy shows that O2 is spring in a symmetric environment (ν(O-O) is not IR-allowed).
- OxyHc is EPR-silent indicating the absence of unpaired electrons
- Infrared spectroscopy shows ν(O-O) of 755 cmâˆ'one
Much work has been devoted to preparing synthetic analogues of the active site of hemocyanin.[xix] I such model, which features a pair of copper centers bridged side-on past peroxo ligand, shows ν(O-O) at 741 cmâˆ'i and a UV-Vis spectrum with absorbances at 349 and 551 nm. Both of these measurements hold with the experimental observations for oxyHc.[xx] The Cu-Cu separation in the model circuitous is three.56 Ã…, that of oxyhemocyanin is ca. iii.6 Ã… (deoxyHc: ca. iv.6 Ã…).[20] [21] [22]
Anticancer furnishings
The hemocyanin found in the claret of the Chilean abalone, Concholepas concholepas, has immunotherapeutic furnishings against float cancer in murine models. Mice primed with C. concholepas before implantation of float tumor (MBT-two) cells. Mice treated with C. concholepas hemocyanin showed antitumor furnishings: prolonged survival, decreased tumor growth and incidence, and lack of toxic furnishings and may have a potential employ in future immunotherapy for superficial bladder cancer.[23]
Keyhole limpet hemocyanin (KLH) is an immune stimulant derived from circulating glycoproteins of the marine mollusk Megathura crenulata. KLH has been shown to be a significant treatment against the proliferations of breast cancer, pancreas cancer, and prostate cancer cells when delivered in vitro. Keyhole limpet hemocyanin inhibits growth of human Barrett's esophageal cancer through both apoptic and nonapoptic mechanisms of cell death.[24]
Case studies: environmental bear upon on hemocyanin levels
A 2003 written report of the effect of culture conditions of blood metabolites and hemocyanin of the white shrimp Litopenaeus vannamei plant that the levels of hemocyanin, oxyhemocyanin in detail, are affected by the nutrition. The study compared oxyhemocyanin levels in the blood of white shrimp housed in an indoor swimming with a commercial diet with that of white shrimp housed in an outdoor swimming with a more than readily available protein source (natural live food) equally well. Oxyhemocyanin and blood glucose levels were higher in shrimp housed in outdoor ponds. It was also found that blood metabolite levels tended to be lower in low activeness level species, such equally crabs, lobsters, and the indoor shrimp when compared to the outdoor shrimp. This correlation is peradventure indicative of the morphological and physiological evolution of crustaceans. The levels of these blood proteins and metabolites appear to exist dependent on energetic demands and availability of those energy sources.[25]
Encounter as well
- Atlantic horseshoe crab blood
- Keyhole limpet hemocyanin
- Hemoglobin
- Myoglobin
- Respiratory pigment
References
- ^ a b Coates CJ, Nairn J (July 2014). "Diverse immune functions of hemocyanins". Developmental and Comparative Immunology. 45 (1): 43â€"55. doi:10.1016/j.dci.2014.01.021. PMID 24486681.
- ^ a b Voit R, Feldmaier-Fuchs G, Schweikardt T, Decker H, Burmester T (Dec 2000). "Complete sequence of the 24-mer hemocyanin of the tarantula Eurypelma californicum. Structure and intramolecular development of the subunits". The Journal of Biological Chemistry. 275 (l): 39339â€"44. doi:10.1074/jbc.M005442200. PMID 10961996.
- ^ Jaenicke Eastward, Pairet B, Hartmann H, Decker H (2012). "Crystallization and preliminary analysis of crystals of the 24-meric hemocyanin of the emperor scorpion (Pandinus imperator)". PLOS I. 7 (3): e32548. Bibcode:2012PLoSO...732548J. doi:10.1371/journal.pone.0032548. PMCÂ3293826. PMIDÂ 22403673.
- "The blue blood of the emperor scorpion x-rayed". Johannes Gutenberg-Universität Mainz. June 22, 2012.
- ^ Beintema JJ, Stam WT, Hazes B, Smidt MP (1994). "Evolution of arthropod hemocyanins and insect storage proteins (hexamerins)". Mol Biol Evol. 11 (iii): 493â€"503. doi:ten.1093/oxfordjournals.molbev.a040129. PMID 8015442.
- ^ Burmester, T (February 2002). "Origin and evolution of arthropod hemocyanins and related proteins". Journal of Comparative Physiology B. 172 (two): 95â€"107. doi:ten.1007/s00360-001-0247-vii. PMID 11916114. S2CID 26023927.
- ^ Lage C, Soderhall K (2004). "The prophenoloxidase-activating system in invertebrates". Immunological Reviews. 198 (1): 116â€"126. doi:x.1111/j.0105-2896.2004.00116.10. PMID 15199959. S2CID 10614298.
- ^ Terwilliger NB (1999). "Hemolymph Proteins and Molting in Crustaceans and Insects". American Zoologist. 39 (3): 589â€"599. doi:x.1093/icb/39.three.589.
- ^ Terwilliger NB, Dangott Fifty, Ryan M (1999). "Cryptocyanin, a crustacean molting protein: Evolutionary link with arthropod hemocyanins and insect hexamerins". PNAS. 96 (5): 2013â€"2018. Bibcode:1999PNAS...96.2013T. doi:ten.1073/pnas.96.5.2013. PMCÂ26728. PMID 10051586.
- ^ Burmester T (February 2001). "Molecular evolution of the arthropod hemocyanin superfamily". Molecular Biology and Evolution. 18 (2): 184â€"95. doi:x.1093/oxfordjournals.molbev.a003792. PMID 11158377.
- ^ Rannulu NS, Rodgers MT (March 2005). "Solvation of copper ions by imidazole: structures and sequential binding energies of Cu+(imidazole)ten, x = one-iv. Contest between ion solvation and hydrogen bonding". Concrete Chemistry Chemical Physics. seven (5): 1014â€"25. Bibcode:2005PCCP....7.1014R. doi:10.1039/b418141g. PMID 19791394.
- ^ Strobel A, Hu MY, Gutowska MA, Lieb B, Lucassen Grand, Melzner F, et al. (December 2012). "Influence of temperature, hypercapnia, and evolution on the relative expression of unlike hemocyanin isoforms in the common cuttlefish Sepia officinalis" (PDF). Journal of Experimental Zoology Part A. 317 (eight): 511â€"23. doi:10.1002/jez.1743. PMID 22791630.
- ^ Sterner R, Vogl T, Hinz HJ, Penz F, Hoff R, Föll R, et al. (1995). "Extreme thermostability of tarantula hemocyanin". FEBS Lett. 364 (ane): 9â€"12. doi:x.1016/0014-5793(95)00341-6. PMID 7750550.
- ^ Perton FG, Beintema JJ, Decker H (May 1997). "Influence of antibody binding on oxygen binding behavior of Panulirus interruptus hemocyanin". FEBS Messages. 408 (ii): 124â€"6. doi:x.1016/S0014-5793(97)00269-X. PMID 9187351.
- ^ Waxman L (May 1975). "The structure of arthropod and mollusc hemocyanins". The Periodical of Biological Chemistry. 250 (10): 3796â€"806. doi:10.1016/S0021-9258(19)41469-5. PMID 1126935.
- ^ a b van Holde KE, Miller KI (1995). "Hemocyanins". In Anfinsen CB, Richards FM, Edsall JT, Eisenberg DS (eds.). Advances in Protein Chemical science. Vol. 47. Bookish Press. pp. 1â€"81. doi:10.1016/S0065-3233(08)60545-8. ISBNÂ978-0-12-034247-1. PMID 8561049.
- ^ Kusche K, Hembach A, Hagner-Holler S, Gebauer W, Burmester T (July 2003). "Consummate subunit sequences, structure and evolution of the half dozen x 6-mer hemocyanin from the mutual business firm centipede, Scutigera coleoptrata". European Journal of Biochemistry. 270 (thirteen): 2860â€"viii. doi:10.1046/j.1432-1033.2003.03664.ten. PMID 12823556.
- ^ a b Decker H, Tuczek F (August 2000). "Tyrosinase/catecholoxidase activity of hemocyanins: structural basis and molecular mechanism" (PDF). Trends in Biochemical Sciences. 25 (8): 392â€"vii. doi:10.1016/S0968-0004(00)01602-nine. PMID 10916160.
- ^ Decker H, Schweikardt T, Nillius D, Salzbrunn U, Jaenicke E, Tuczek F (August 2007). "Like enzyme activation and catalysis in hemocyanins and tyrosinases". Gene. 398 (1â€"2): 183â€"91. doi:10.1016/j.gene.2007.02.051. PMID 17566671.
- ^ a b Elwell CE, Gagnon NL, Neisen BD, Dhar D, Spaeth AD, Yee GM, et al. (2017). "Copperâ€"Oxygen Complexes Revisited: Structures, Spectroscopy, and Reactivity". Chemical Reviews. 117 (3): 2059â€"2107. doi:x.1021/acs.chemrev.6b00636. PMCÂ5963733. PMID 28103018.
- ^ a b Kitajima Due north, Fujisawa Yard, Fujimoto C, Morooka Y, Hashimoto S, Kitagawa T, et al. (1992). "A new model for dioxygen binding in hemocyanin. Synthesis, characterization, and molecular structure of the μ-η2:η2 peroxo dinuclear copper(II) complexes, [Cu(BH(three,5-R2pz)three)]2(O2) (R = i-Pr and Ph)". Periodical of the American Chemical Society. 114 (4): 1277â€"91. doi:10.1021/ja00030a025.
- ^ Gaykema WP, Hol WG, Vereijken JM, Soeter NM, Bak HJ, Beintema JJ (1984). "3.2 Ã… structure of the copper-containing, oxygen-carrying poly peptide Panulirus interruptus haemocyanin". Nature. 309 (5963): 23â€"9. Bibcode:1984Natur.309...23G. doi:x.1038/309023a0. S2CID 4260701.
- ^ Kodera K, Katayama K, Tachi Y, Kano K, Hirota Due south, Fujinami S, et al. (1999). "Crystal Structure and Reversible O2-Binding of a Room Temperature Stable μ-η2:ηii-Peroxodicopper(II) Circuitous of a Sterically Hindered Hexapyridine Dinucleating Ligand". Journal of the American Chemic Society. 121 (47): 11006â€"7. doi:10.1021/ja992295q.
- ^ Atala A (2006). "This Calendar month in Investigative Urology". The Journal of Urology. 176 (6): 2335â€"6. doi:10.1016/j.juro.2006.09.002.
- ^ McFadden DW, Riggs DR, Jackson BJ, Vona-Davis 50 (November 2003). "Keyhole limpet hemocyanin, a novel allowed stimulant with promising anticancer activity in Barrett's esophageal adenocarcinoma". American Journal of Surgery. 186 (5): 552â€"v. doi:x.1016/j.amjsurg.2003.08.002. PMID 14599624.
- ^ Pascual C, Gaxiola G, Rosas C (2003). "Claret metabolites and hemocyanin of the white shrimp, Litopenaeus vannamei: The upshot of civilisation conditions and a comparison with other crustacean species". Marine Biology. 142 (4): 735. doi:ten.1007/s00227-002-0995-2. S2CIDÂ 82961592.
Further reading
- Rehm P, Pick C, Borner J, Markl J, Burmester T (February 2012). "The diverseness and evolution of chelicerate hemocyanins". BMC Evolutionary Biological science. 12: 19. doi:10.1186/1471-2148-12-nineteen. PMCÂ3306762. PMIDÂ 22333134.
- Ali SA, Abbasi A (2011). Scorpion Hemocyanin: The bluish claret. Saarbrücken: VDM Verlag Dr. Müller. p. 160. ISBNÂ978-iii-639-33725-nine.
External links
| | Wikimedia Commons has media related to Hemocyanin. |
- 3D hemocyanin structures in the EM Information Banking concern (EMDB)
- Overview of all the structural information available in the PDB for UniProt: P04253 (Hemocyanin 2) at the PDBe-KB.
This page is based on a Wikipedia article. The text is bachelor nether the Creative Eatables Attribution/Share-Alike License.
This tab holds the annotation information that is stored in the Pfam database. Equally nosotros move to using Wikipedia as our main source of annotation, the contents of this tab will be gradually replaced past the Wikipedia tab.
Hemocyanin, all-alpha domain Provide feedback
This family includes arthropod hemocyanins and insect larval storage proteins.
Literature references
-
Jones One thousand, Dark-brown Due north, Manczak M, Hiremath Southward, Kafatos FC; , J Biol Chem 1990;265:8596-8602.: Molecular cloning, regulation, and complete sequence of a hemocyanin-related, juvenile hormone-suppressible poly peptide from insect hemolymph. PUBMED:2341396 EPMC:2341396
-
Willott E, Wang XY, Wells MA; , J Biol Chem 1989;264:19052-19059.: cDNA and gene sequence of Manduca sexta arylphorin, an aromatic amino acid-rich larval serum protein. Homology to arthropod hemocyanins. PUBMED:2808410 EPMC:2808410
-
Hazes B, Magnus KA, Bonaventura C, Bonaventura J, Dauter Z, Kalk KH, Hol WG; , Protein Sci 1993;ii:597-619.: Crystal structure of deoxygenated Limulus polyphemus subunit 2 hemocyanin at 2.xviii A resolution: clues for a mechanism for allosteric regulation. PUBMED:8518732 EPMC:8518732
External database links
| HOMSTRAD: | hemocyan |
| PRINTS: | PR00187 |
| PROSITE: | PDOC00184 |
| SCOP: | 1lla |
This tab holds note data from the InterPro database.
InterPro entry IPR005204
Crustacean and cheliceratan hemocyanins (oxygen-transport proteins) and insect hexamerins (storage proteins) are homologous cistron products, although the latter do non bind oxygen [ PUBMED:8015442 ].
Haemocyanins are found in the haemolymph of many invertebrates. They are divided into 2 primary groups, arthropodan and molluscan. These have structurally similar oxygen-binding centres, which are like to the oxygen-binding centre of tyrosinases, merely their quaternary structures are arranged differently. The arthropodan proteins exist as hexamers comprising 3 heterogeneous subunits (a, b and c) and possess one oxygen-binding centre per subunit; and the molluscan proteins exist as cylindrical oligomers of 10 to 20 subunits and possess 7 or 8 oxygen-binding centres per subunit [ PUBMED:3207675 ]. Although the proteins have like amino acid compositions, the simply real similarity in their primary sequences is in the region corresponding to the second copper-binding domain, which also shows similarity to the copper-binding domain of tyrosinases.
Hexamerins are proteins from the hemolymph of insects, which may serve as a store of amino acids for synthesis of adult proteins. They do not possess the copper-binding histidines present in hemocyanins [ PUBMED:8015442 ].
Homologues are likewise present in other kinds of organism, for instance, AsqI from the yeast Emericella nidulans. This is a tyrosinase involved in biosynthesis of the aspoquinolone mycotoxins, though its exact function is unknown [ PUBMED:25251934 ].
This entry represents the Due north-terminal domain of hemocyanin and hexamerin proteins.
Source: https://pfam.xfam.org/family/PF03722
0 Response to "what is the maximum number of o_2 2 ​ molecules that can bind to a single molecule of hemoglobin?"
Post a Comment